In vertebrates, the mesoderm of the head is unsegmented compared to that of the trunk, which forms repeated structures called somites. In amphioxus, all non-axial mesoderm is segmented into somites, and this trait is proposed as ancestral to chordates. This implies that muscles derived from vertebrate head mesoderm would be a character specific to this lineage. Through the study of somitogenesis,of the transcription factors involved in this process, and through the use of scRNA-seq approaches (in collaboration with the team of Arnau Sebé-Pedrós, CRG, Barcelona), we have proposed a new evolutionary scenario for the appearance of vertebrate head muscles. This involves not only the evolution of somitogenesis, but also the evolution of different mesodermal territories, including the prechordal plate and the lateral mesoderm. This project is funded by the ANR (MESOLAMPHIX project) and is being developed in collaboration with the teams of Sylvie Mazan (BIOM), Juan Tena (CABD, Seville) and Arnau Sebé-Pedrós (CRG, Barcelona).
Key publications :
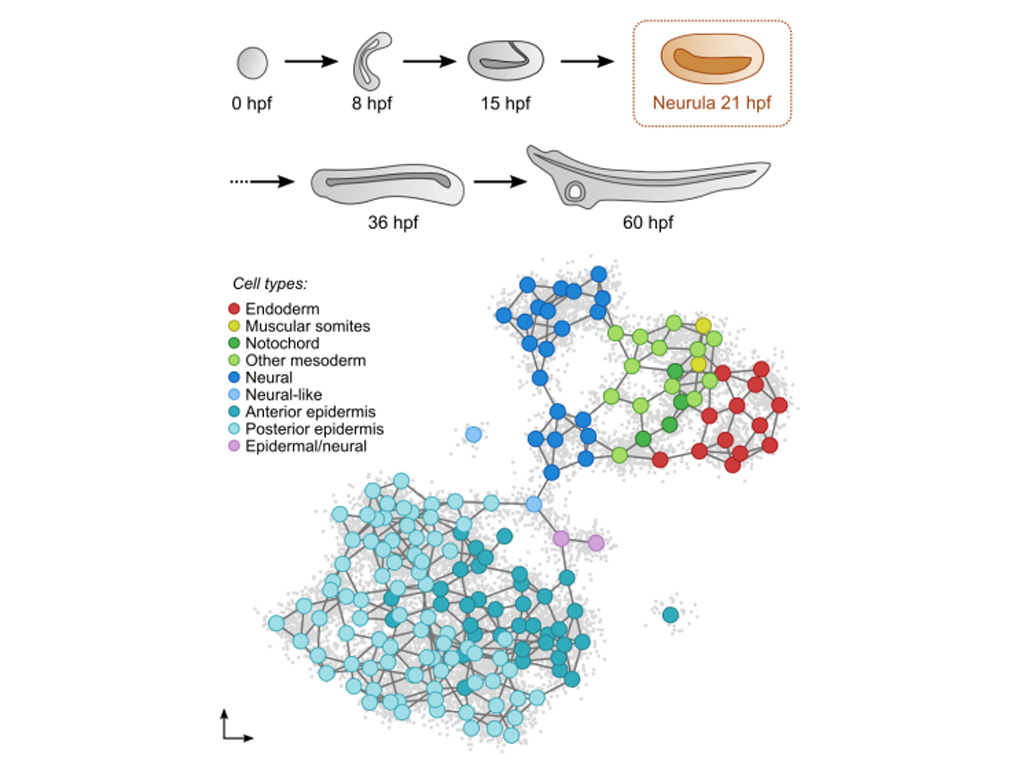
Grau-Bové X**, Subirana L**, Meister L, Soubigou A, Neto A, Elek A, Naranjo S, Fornas O, Gomez-Skarmeta JL, Tena JJ, Irimia M, Bertrand S*, Sebé-Pedrós A*, Escriva H*. An amphioxus neurula stage cell atlas supports a complex scenario for the emergence of vertebrate head mesoderm. Nat Commun. 2024 May 29;15(1):4550. https://doi.org/10.1038/s41467-024-48774-4
Meister L, Escriva H*, Bertrand S*. Functions of the FGF signalling pathway in cephalochordates provide insight into the evolution of the prechordal plate. Development. 2022 May 15;149(10):dev200252. https://doi.org/10.1242/dev.200252
Aldea D, Subirana L, Keime C, Meister L, Maeso I, Marcellini S, Gomez-Skarmeta JL, Bertrand S*, Escriva H*. Genetic regulation of amphioxus somitogenesis informs the evolution of the vertebrate head mesoderm. Nat Ecol Evol. 2019 Aug;3(8):1233-1240. https://doi.org/10.1038/s41559-019-0933-z
Bertrand S, Camasses A, Somorjai I, Belgacem MR, Chabrol O, Escande ML, Pontarotti P, Escriva H. Amphioxus FGF signaling predicts the acquisition of vertebrate morphological traits. Proc Natl Acad Sci U S A. 2011 May 31;108(22):9160-5. https://doi.org/10.1073/pnas.1014235108
**contributed equally
*co-corresponding authors
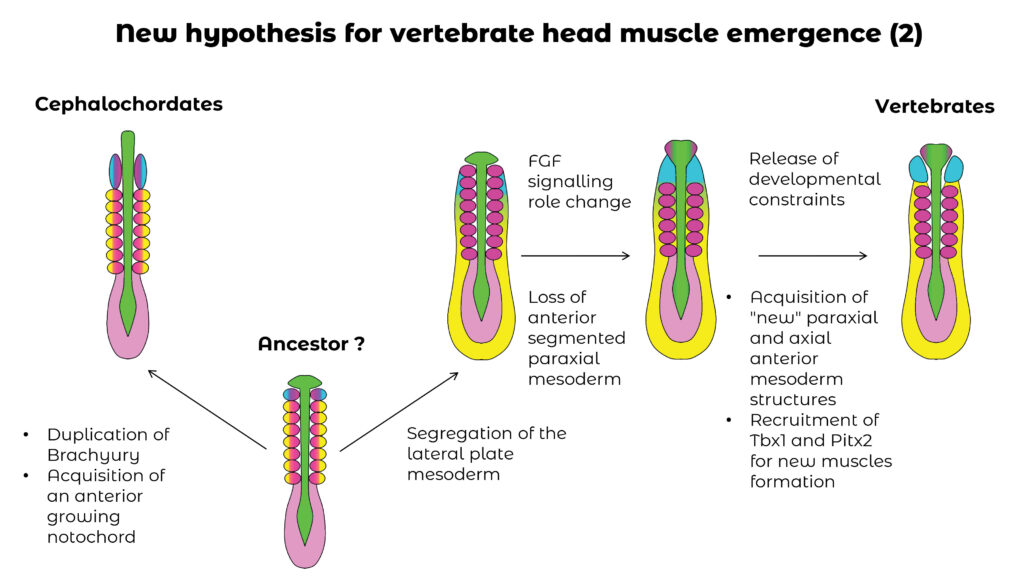
Scenario for the evolution of the different compartments of the mesoderm in chordates. We propose that several stages were necessary for the appearance of vertebrate head muscles.
Vertebrate head muscles appear to be an innovation of this group (see project 1) and we have proposed an evolutionary scenario for their emergence (Figure 1). The activity of these muscles is controlled by specific motor neurons, different from those controlling the muscles originating from the somites (trunk and limbs). The aim of the project is to determine whether these neurons are homologous to motoneurons controlling anterior (oropharyngeal) muscle activity in the other two groups of chordates: tunicates and cephalochordates. This project is funded by the ANR (Projet COMUNE) and is being developed in collaboration with the teams of Jean-François Brunet (IBENS, Paris), Sébastien Darras (BIOM) and Arnau Sebé-Pedrós (CRG, Barcelona).
Neural induction is the process by which pluripotent cells of the ectoderm acquire a neural fate. We have shown that this process in amphioxus, as in vertebrates, involves a particular region of the embryo known as the “organizer”. We have also demonstrated that this process is under the control of the Nodal signaling pathway. Finally, we were able to dissect the gene regulatory network controlling neural induction. The current aim is to use scRNA-seq approaches to better define the key players in developmental trajectory transitions during neural induction. The project was funded by the ANR (NEUCECHO project) in collaboration with the teams of Jose Luis Gomez-Skarmeta and Juan Tena (CABD, Seville) and of Arnau Sebé-Pedrós (CRG, Barcelona).
Key publications :
Leon A, Subirana L, Magre K, Cases I, Tena JJ, Irimia M, Gomez-Skarmeta JL, Escriva H*, Bertrand S*. Gene Regulatory Networks of Epidermal and Neural Fate Choice in a Chordate. Mol Biol Evol. 2022 Apr 11;39(4):msac055. https://doi.org/10.1093/molbev/msac055
Le Petillon Y, Luxardi G, Scerbo P, Cibois M, Leon A, Subirana L, Irimia M, Kodjabachian L, Escriva H*, Bertrand S*. (2017) Nodal/Activin Pathway is a Conserved Neural Induction Signal in Chordates. Nat Ecol Evol. 2017 Aug;1(8):1192-1200. https://doi.org/10.1038/s41559-017-0226-3
*co-corresponding authors
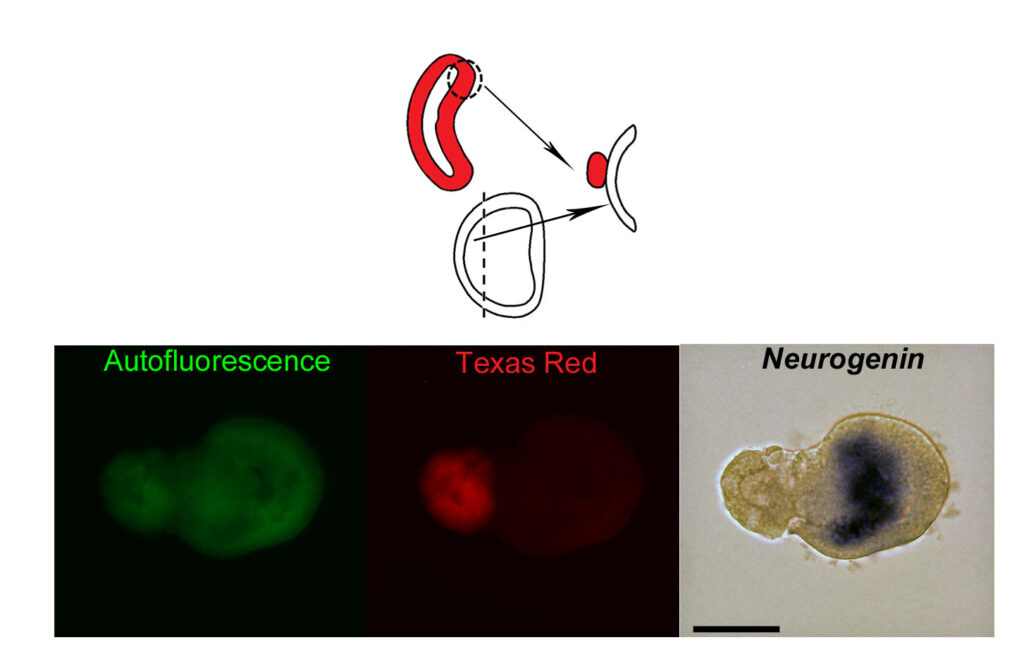
The dorsal lip of the blastopore of a gastrula after injection of TexasRed was grafted onto an ectodermal explant developing into epidermis when cultured alone. The host shows expression of the neural marker Neurogenin, indicating that the graft (red fluorescent) has induced the acquisition of neural fate by cells that normally enter the epidermal differentiation program. Scale bar: 50µM. Modified from: Le Petillon Y et al., Nodal/Activin Pathway is a Conserved Neural Induction Signal in Chordates. Nat Ecol Evol. 2017 Aug;1(8):1192-1200
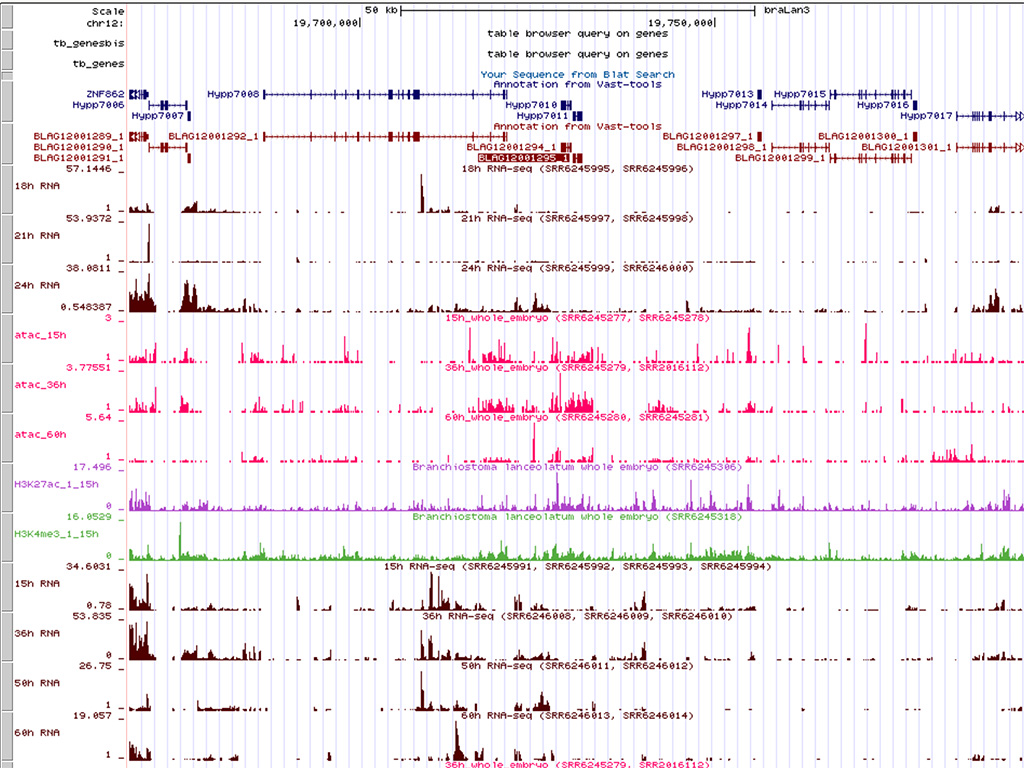
Our team is a leader in developmental studies in amphioxus. Our aim is to continue developing new tools and techniques. We have obtained the complete genome sequence and the chromosome scale assembly of the European species Branchiostoma lanceolatum, as well as numerous epigenomic data in collaboration with Génoscope and several international laboratories. In collaboration with several teams, we have developed RNA-seq, ATAC-seq, ChIP-seq and scRNA-seq techniques, and more recently mutagenesis using the CRISPR/Cas9 system (in collaboration with Sébastien Darras, BIOM) and cell tracing after photoconversion.
Key publications :
D’Aniello S, Bertrand S, Escriva H. Amphioxus as a model to study the evolution of development in chordates. Elife. 2023 Sep 18;12:e87028. https://doi.org/10.7554/eLife.87028
Brasó-Vives M, Marlétaz F, Echchiki A, Mantica F, Acemel RD, Gómez-Skarmeta JL, Hartasánchez DA, Le Targa L, Pontarotti P, Tena JJ, Maeso I, Escriva H, Irimia M, Robinson-Rechavi M. Parallel evolution of amphioxus and vertebrate small-scale gene duplications. Genome Biol. 2022 Nov 18;23(1):243.. https://doi.org/10.1186/s13059-022-02808-6
Meister L, Escriva H*, Bertrand S*. Exploring tissue morphodynamics using the photoconvertible Kaede protein in amphioxus embryos. PLoS One. 2022 Sep 27;17(9):e0275193. https://doi.org/10.1371/journal.pone.0275193
Escriva H. (2018). My Favorite Animal, Amphioxus: Unparalleled for Studying Early
Escriva H. My Favorite Animal, Amphioxus: Unparalleled for Studying Early Vertebrate Evolution. Bioessays. 2018:e1800130. doi: 10.1002/bies.201800130
Marlétaz F, Firbas PN, Maeso I, Tena JJ, Bogdanovic O, Perry M, Wyatt CDR, de la Calle-Mustienes E, Bertrand S, Burguera D, Acemel RD, van Heeringen SJ, Naranjo S, Herrera-Ubeda C, Skvortsova K, Jimenez-Gancedo S, Aldea D, Marquez Y, Buono L, Kozmikova I, Permanyer J, Louis A, Albuixech-Crespo B, Le Petillon Y, Leon A, Subirana L, Balwierz PJ, Duckett PE, Farahani E, Aury JM, Mangenot S, Wincker P, Albalat R, Benito-Gutiérrez È, Cañestro C, Castro F, D’Aniello S, Ferrier DEK, Huang S, Laudet V, Marais GAB, Pontarotti P, Schubert M, Seitz H, Somorjai I, Takahashi T, Mirabeau O, Xu A, Yu JK, Carninci P, Martinez-Morales JR, Crollius HR, Kozmik Z, Weirauch MT, Garcia-Fernàndez J, Lister R, Lenhard B, Holland PWH, Escriva H*, Gómez-Skarmeta JL*, Irimia M*. (2018). Amphioxus functional genomics and the origins of vertebrate gene regulation. Nature. 2018Dec;564(7734):64-70. https://doi.org/10.1038/s41586-018-0734-6
Bertrand S, Escriva H. (2011). Evolutionary crossroads in developmental biology: amphioxus. Development. 2011 Nov;138(22):4819-30. https://doi.org/10.1242/dev.066720
*co-corresponding authors
.
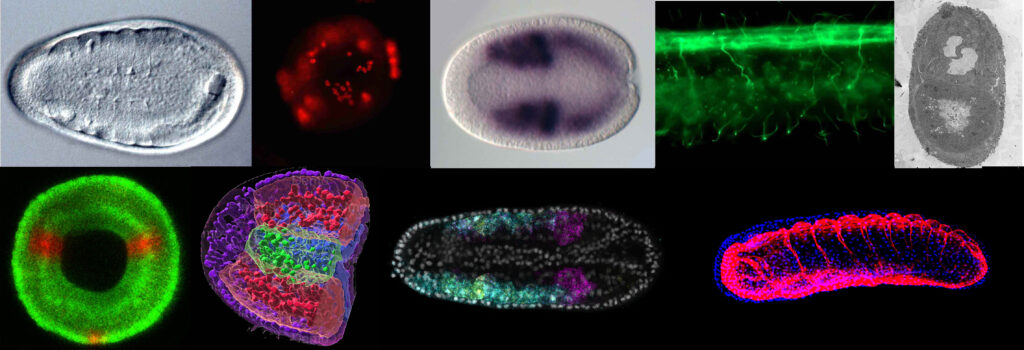
Amphioxus from every angle. From left to right, top to bottom: amphioxus neurula under light microscope, gastrula after immunostaining for phosphoH3, neurula after colorimetric in situ hybridization (Delta), region of an amphioxus larva after immunostaining for acetylated tubulin, electron microscopic view of a larval cross-section, blastoporal view of a gastrula after photoconversion of kaede protein, partial 3D reconstruction of a neurula after cell segmentation, neurula after in situ hybridization with the HCR method for three genes, 3D view of a late neurula after labeling of nuclei (blue) and basal lamina (red).